Industry
Healthcare
Client
Ebenbuild — health tech startup, €4.8M raised
Increasing survival rates of patients with acute respiratory distress by creating a digital lung “twin” (0→1)
WHAT — Medical device software helping doctors treat acute respiratory distress (ARDS) WHY — No intuitive tools existed for life-critical ICU ventilator workflows HOW — Led design strategy, user research, and design system, from prototype to clinical trials IMPACT — Raised €4.8M from the European Innovation Council and Bayern Kapital. Up to €10M in additional equity financing to follow. Immediate value for fundraising and partnerships.

Leading design from funded prototype to clinical-ready product
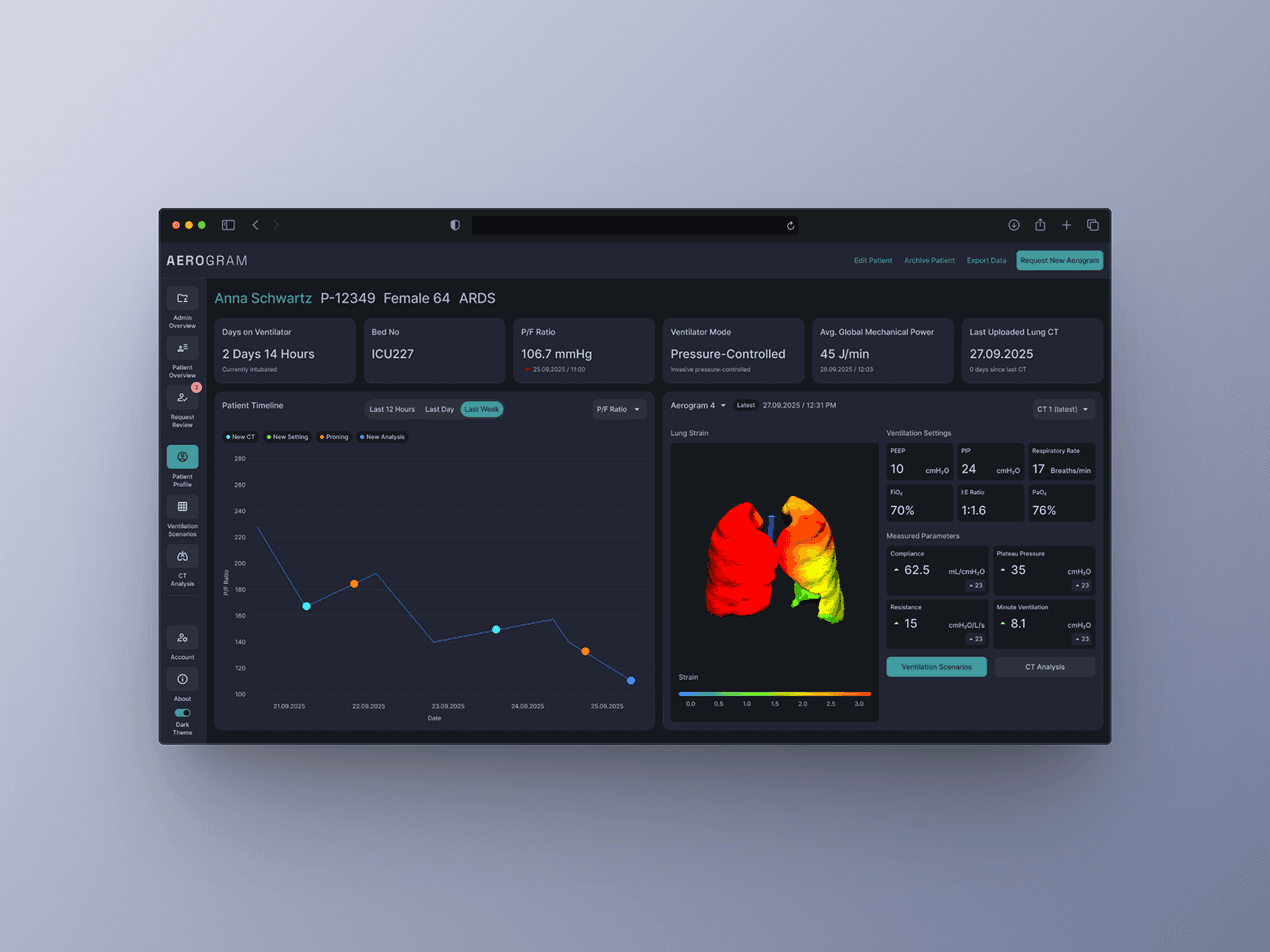
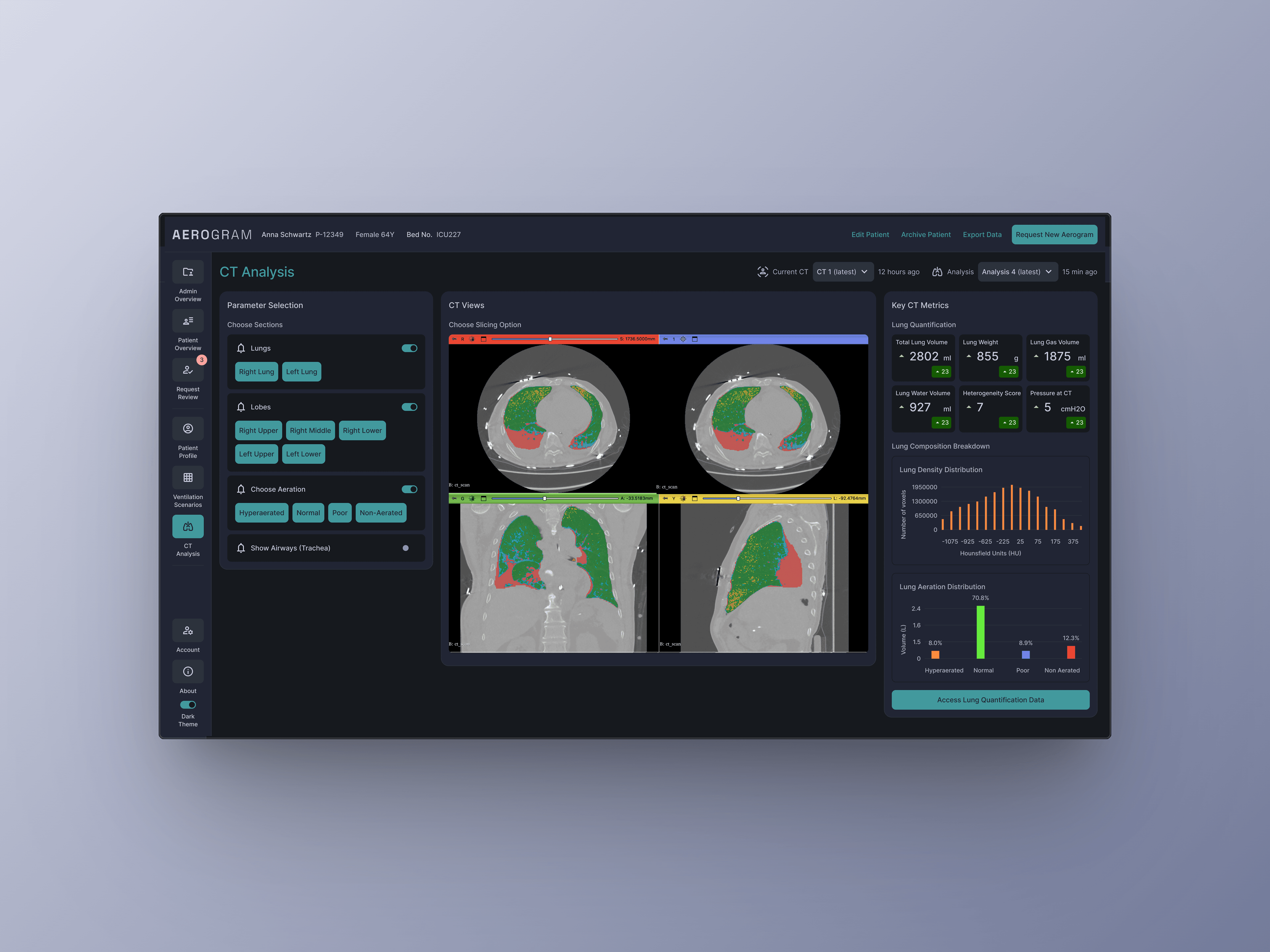
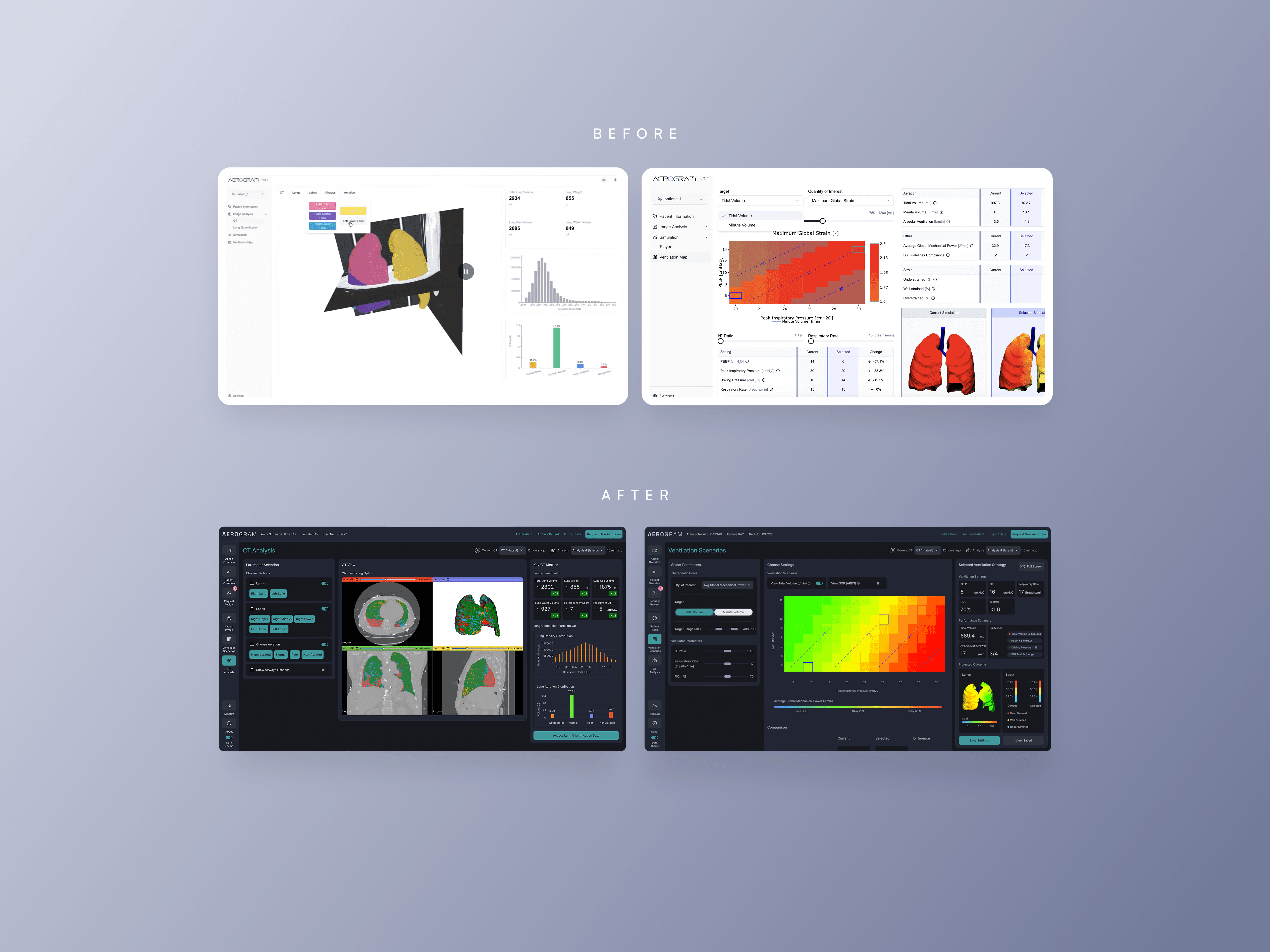
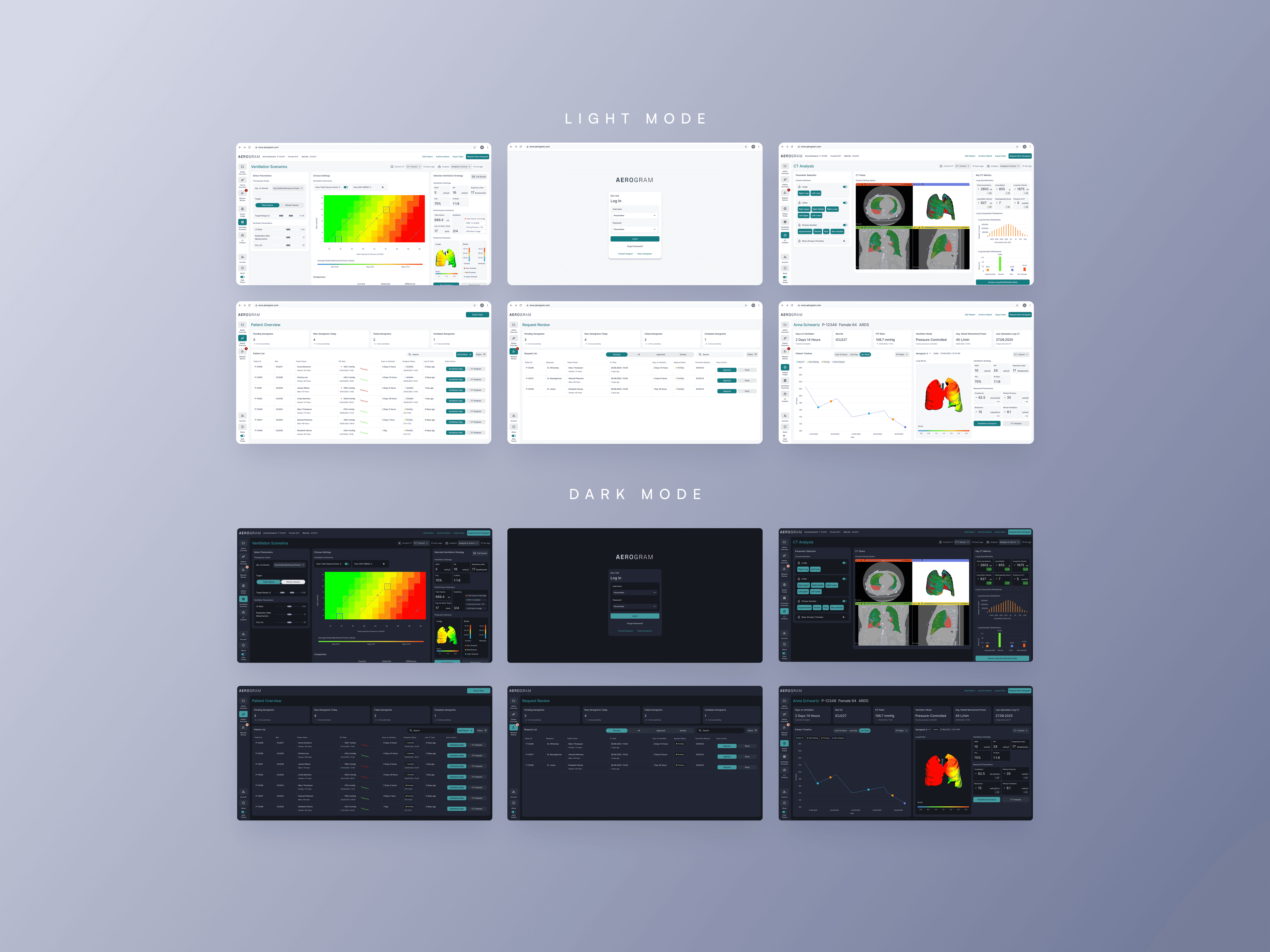
CHALLENGE: Ebenbuild's digital lung twin had funding and validation, but the engineer-built prototype wasn't grounded in real clinical workflows. With no comparable tool on the market and a future as an FDA-regulated medical device, we needed to build something intuitive enough for high-pressure ICU environments - where bad UX could mean the difference between adoption and abandonment. MY ROLE: I led design strategy and execution with a visual designer, working directly with the PM, Engineering and C-level team. APPROACH: I conducted extensive on-site and remote research and testing with clinicians in hospitals across Germany and the US, and along with a visual design lead - helped build a design system optimized for clinical clarity. I bridged clinical needs, technical constraints, and business goals - bringing stakeholders together at key moments to align on direction. Even though branding wasn't a part of the brief, I went the extra mile to think about a brand story and come up with a visual identity that helps the tool stand out and be noticeable in a hospital setting through communication materials.
The result: A first-of-its-kind tool that clinicians actually want to use
“Radically new technology such as ours is toothless if no one wants to use it and very few can already imagine its possibilities. Daylight has made these possibilities remarkably tangible and has already sparked people’s imagination.” – Dr. Kei W. Müller Co-Founder and CEO at Ebenbuild GmbH “I think this tool will really help us improve outcomes if we can build it into the workflow.” - Dr. Sebastian Billig, Assistenzarzt Uniklinik RWTH Aachen
We delivered a modern, intuitive tool that clinicians were genuinely excited about. The product is currently going through clinical trials, on track for market launch in 2026 as an FDA-regulated medical device. More importantly, we built a design foundation that can scale as the product evolves - balancing regulatory rigor with human-centered design. The work proved that even in highly technical, regulated contexts, thoughtful design is what earns trust and drives adoption.
Daylight Team: Siddharth Dasari (Project and Client Lead, UX-Visual Design, Design Research), David Hwang (Visual Design Lead), Johanna Gieseler (Research Support), Jeremy Borden (Engineering) Ebenbuild Team: Nina Pischke (PM), Max-Karl Wachter (Engineering), Jonas Biehler (CTO), Kai Muller (CEO)
AI Experiments